We know the atomic number of hydrogen is one, so there's one proton in the nucleus. So let's go ahead and draw an atom of hydrogen. In a neutral atom, the number of protons is equal to the number of electrons, because in a neutral atom there's no overall charge and the positive charges of the protons completely balance with the negative charges of the electrons. So it's right here, so there's one proton in the nucleus of a hydrogen atom. 
So for hydrogen, hydrogen's atomic number is one. So we're going to talk about hydrogen in this video. And you can find the atomic number on the periodic table. So the atomic number is symbolized by Z and it refers to the number of protons in a nucleus. There are ways it can happen, but they are not the normal course of events. So you do not usually have one isotope changing into another isotope of the same element. So, it is not possible to give you a general answer other than to say that the isotopes of an element form in one or more of the above ways, but each isotope of an element may or may not form in a similar way as other isotopes.Įxcept as listed above, an atom that is not radioactive never changes its number of neutrons. There are a few other ways, but they are not all that significant.Įach isotope (more properly called a nuclide) has its own way(s) in which it is formed. Being changed from one type of atom to another by high energy nuclear reactions, such as having a neutron slam into the nucleus at an extremely high velocity. When an electric field is applied, the ions are accelerated into a separate chamber where they are deflected from their initial trajectory by a magnetic field, like the electrons in Thomson’s experiment.With the exception of the hydrogen and helium that were formed shortly after the Big Bang event, elements mainly form in the following ways:Ĥ. First, electrons are removed from or added to atoms or molecules, thus producing charged particles called ions. The technique is conceptually similar to the one Thomson used to determine the mass-to-charge ratio of the electron. Scientists can measure relative atomic masses very accurately, however, using an instrument called a mass spectrometer. Although the difference in mass is small, it is extremely important because it is the source of the huge amounts of energy released in nuclear reactions.īecause atoms are much too small to measure individually and do not have charges, there is no convenient way to accurately measure absolute atomic masses. For example, the ratio of the masses of 1H (hydrogen) and 2H (deuterium) is actually 0.500384, rather than 0.49979 as predicted from the numbers of neutrons and protons present. Once the masses of atoms were determined, the amu could be assigned an actual value:ġ amu = 1.66054 x 10 -24 grams conversely:Īlthough the masses of the electron, the proton, and the neutron are known to a high degree of precision ( Table 2.3.1), the mass of any given atom is not simply the sum of the masses of its electrons, protons, and neutrons. Thus, the mass of the hydrogen atom ( 1H) is 1.0080 amu, and the mass of an oxygen atom ( 16O) is 15.995 amu. The atomic mass unit ( amu) was not standardized against hydrogen, but rather, against the 12C isotope of carbon ( amu = 12). 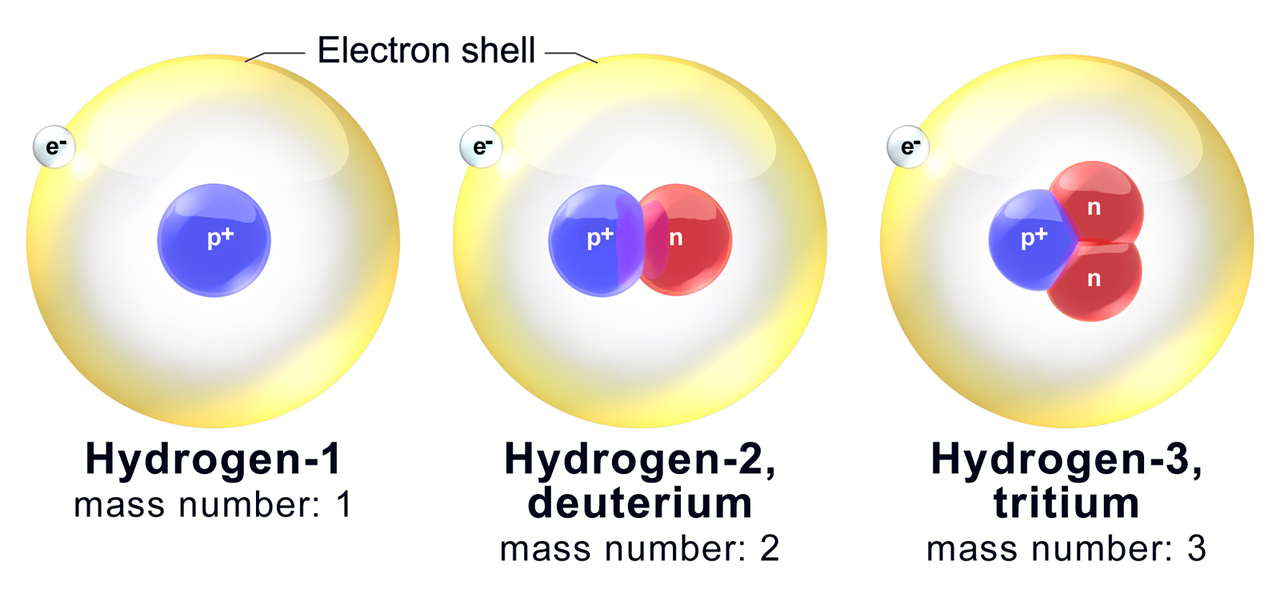
As we saw earlier, it is convenient to use a reference unit when dealing with such small numbers: the atomic mass unit. We now know that a hydrogen atom has a mass of 1.6735 x 10 -24 grams, and that the oxygen atom has a mass of 2.6561 X 10 -23 grams. Thus, oxygen was assigned an atomic mass of 16. Hydrogen, the lightest element, was assigned a relative mass of '1', and the other elements were assigned 'atomic masses' relative to this value for hydrogen.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
